Executive Summary
The rapid advancement of modern drugs and biologics, including small molecules, peptides, proteins, and nucleic acids, has outpaced conventional delivery technologies. Synthetic lipids have emerged as a versatile platform for efficient, biocompatible delivery of diverse therapeutic payloads.
Engineered for precise structure and performance, synthetic lipids outperform natural lipids in stability, reproducibility, payload compatibility, and control of biodistribution and release, supporting systems such as lipid nanoparticles, liposomes, emulsions, and lipid-polymer hybrids.
Synthetic lipid delivery systems are now applied across oncology, rare and infectious diseases, vaccines, and localized routes including ocular and pulmonary administration. This white paper outlines their scientific foundation, applications, and strategic importance in next-generation drug and biologics delivery.
Introduction
Rapid advances in drug and biologic development have intensified the challenge of effective delivery, as many therapeutics suffer from instability, poor targeting, and limited cellular uptake. Engineered delivery systems are therefore essential to achieve clinical efficacy.
Lipid-based platforms such as liposomes and lipid nanoparticles offer biocompatibility and payload versatility but are often limited by the stability and variability of naturally derived lipids.
Synthetic lipids overcome these constraints through precise structural control, enabling optimized pharmacokinetics, biodistribution, and payload release. This white paper examines their design, applications, and clinical relevance as enabling technologies for next-generation drug and biologics delivery.
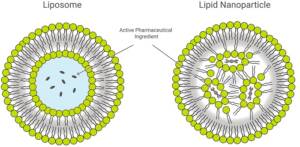
Figure: Liposome & Lipid Nanoparticle
Challenges in Drug, Biologic & Gene Delivery
Many modern therapeutics face fundamental delivery limitations that restrict clinical efficacy. Poor solubility and instability lead to rapid degradation, while non-specific distribution increases off-target toxicity and limits dose escalation. Large molecular size, negative charge, and biological barriers impede tissue penetration and cellular uptake, particularly for nucleic acids such as mRNA, which are highly susceptible to enzymatic degradation. Additional challenges include inefficient endosomal escape and difficulty crossing specialized barriers such as the blood–brain barrier, ocular tissues, and pulmonary epithelium.
Advantages of Lipid-Based Delivery Systems
Lipid-based delivery systems address these challenges through biocompatible and biodegradable architectures that protect therapeutic payloads and enhance stability. Liposomes and lipid nanoparticles improve pharmacokinetics, enable targeted cellular uptake, and reduce systemic toxicity. Their modular design allows surface modification with targeting ligands to support receptor-mediated transport and barrier penetration. While natural lipids offer inherent biocompatibility, synthetic lipids provide superior structural control, stability, and scalability, making them better suited for advanced drug and biologics delivery.
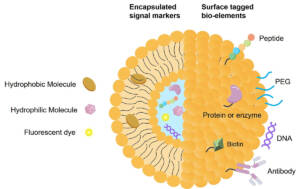
Figure: Targeted NDDS
Synthetic Lipids
Synthetic lipids are purpose-engineered molecules designed to improve the performance of drug and biologic delivery systems beyond naturally derived lipids. Their modular composition enables compatibility with diverse therapeutic modalities, making them a core platform for next-generation delivery.
- Structural Tunability: Customizable headgroups, linkers, and tails enable precise control of charge and membrane interactions.
- Enhanced Stability: Protect APIs from enzymatic, pH, and hydrolytic degradation while improving circulation time.
- Controlled Release: Support sustained or stimuli-responsive payload release for optimized pharmacokinetics.
- High Biocompatibility: Less immunogenicity through biodegradable designs and optimized ionization.
- Efficient Delivery: Facilitate biological barrier penetration and endosomal escape.
- Targeting Flexibility: Surface modification with ligands or PEG enables tissue- and receptor-specific delivery.
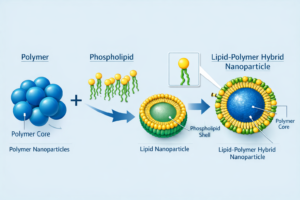
Figure 3: Lipid-Polymer Hybrid Nanoparticle
Types of Synthetic Lipids
-
Phospholipids
Primary Role: Structural integrity, bilayer formation, and membrane mimicry.
Key Characteristics:
- Define bilayer rigidity, fluidity, and phase transition temperature (Tm)
- Contribute to mechanical stability and biocompatibility
- Influence release behavior and formulation robustness
Representative Examples:
- DSPC: High Tm lipid forming rigid, highly stable bilayers.
- DPPC: Pulmonary surfactant analog with moderate Tm.
- DMPC: Forms fluid bilayers at physiological temperatures.
- HSPC: Hydrogenated soy phosphatidylcholine with excellent stability in parenteral liposomal formulations.
-
Cationic Lipids
Primary Role: Electrostatic complexation with nucleic acids and enhancement of cellular uptake.
Key Characteristics:
- Permanently positively charged headgroups
- Strong interaction with cell membranes and nucleic acids
- High transfection efficiency, with toxicity limiting systemic use
Representative Examples:
- DOTAP: Widely used liposome forming lipid for gene delivery.
- DODAP: pH-responsive cationic lipid with reduced charge at physiological pH.
- DSTAP: Sterol-based cationic lipid offering improved serum stability.
-
Anionic Lipids
Primary Role: Surface charge modulation, membrane stability, and biomimetic formulation design.
Key Characteristics:
- Negatively charged headgroups supporting electrostatic balance and particle stability
- Reduced nonspecific interactions and favorable tolerability
- Influence biodistribution and immune response
Representative Examples:
- Phosphatidylglycerols (PG): Stabilize lipid assemblies and modulate surface charge.
- Phosphatidylserines (PS): Support biomimetic and immunomodulatory delivery strategies.
- Phosphatidic acid (PA): Contributes to membrane curvature and structural integrity.
-
PEGylated Lipids (PEG-Lipids)
Primary Role: Stabilization of lipid particles and extension of systemic circulation.
Key Characteristics:
- Hydrophilic PEG chains provide steric shielding against opsonization
- Tunable PEG length and lipid anchors control circulation half-life
- Enable surface functionalization and targeting strategies
Representative Examples:
- DSPE-PEG2000: Standard PEG-lipid for LNPs and stealth liposomes with excellent biocompatibility.
- DMG-PEG2000: Shorter in vivo residence, enabling transient shielding and controlled deshielding.
- Cholesterol PEG conjugates: Enhance membrane anchoring while maintaining steric stabilization.
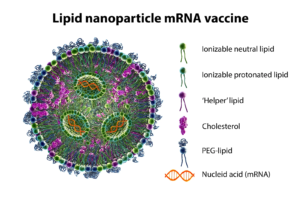
Figure: Composition of Lipid Nanoparticles (LNPs)
Encapsulation Strategies
Synthetic lipids enable efficient drug and biologic delivery by self-assembling into nanoscale carriers that encapsulate therapeutic payloads and transport them to target cells.
- Hydrophilic therapeutics (e.g., peptides, small proteins, select chemotherapeutics) are typically encapsulated within the aqueous core of liposomes or lipid nanoparticles (LNPs).
- Hydrophobic drugs (e.g., paclitaxel, vincristine, steroids) partition into the lipid bilayer or solid lipid matrix, improving solubility and formulation stability.
- Nucleic acids (e.g., mRNA, siRNA, plasmid DNA) form electrostatic complexes with cationic or ionizable lipids, protecting them from enzymatic degradation and enabling intracellular delivery.
Encapsulation is commonly achieved during nanoparticle formation using techniques such as ethanol injection, microfluidic mixing, or thin-film hydration, where lipid components spontaneously self-assemble around the therapeutic payload.
Delivery Pathways
Liposomes and lipid nanoparticles share key biophysical properties with cellular membranes, enabling their nanoscale carriers to interact with cells through multiple, often concurrent, uptake pathways.
- Receptor-mediated interactions with membrane associated proteins or surface components facilitate selective cellular recognition and uptake.
- Endocytic uptake, including clathrin mediated, caveolae mediated, and phagocytic pathways, enables internalization of lipid carriers into endosomal compartments.
- Direct membrane fusion allows lipid components of the nanoparticle to merge with the plasma membrane, releasing the encapsulated payload directly into the cytosol.
- Lipid exchange mechanisms permit transfer of lipid components between the nanoparticle and the cell membrane, influencing membrane composition and payload release.
The dominant pathway depends on the nanoparticle composition, surface chemistry & target cell type; multiple mechanisms may operate simultaneously.
Through precise molecular design, synthetic lipids provide control across each stage of delivery, from cellular interaction and uptake to intracellular release, making them essential to advanced therapies such as gene editing, mRNA vaccines, targeted oncology, and ocular drug delivery.
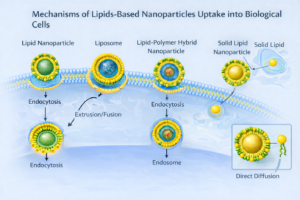
Figure 5: Mechanisms of Nanoparticle uptake into cells
Applications in Drugs and Biologics Delivery
1. Nucleic Acid Delivery:
Synthetic lipid nanoparticles enable delivery of mRNA, siRNA, plasmid DNA, and gene-editing components by protecting them from degradation and promoting endosomal escape. Clinically proven examples include Comirnaty and Spikevax (mRNA vaccines) and Onpattro (siRNA therapy).
2. Protein and Peptide Delivery:
Lipid carriers protect proteins and peptides from proteolysis while preserving bioactivity and enabling sustained or intracellular delivery. Examples include liposomal recombinant enzymes and peptide formulations such as insulin and lactoferrin liposomes in clinical development.
3. Small Molecule Solubilization & Enhancement:
Lipid systems enhance solubility, stability, and bioavailability of hydrophobic APIs while enabling controlled release. Approved examples include Doxil, AmBisome, and DepoDur.
4. Ocular Delivery:
Lipid formulations improve ocular residence time, permeability, and tolerability for anterior and posterior segment delivery. Investigational examples include liposomal cyclosporine for dry eye disease.
5. Pulmonary and Inhaled Therapies:
Surfactant-compatible lipid nanoparticles enable stable aerosol delivery and localized lung targeting. Examples include DPPC/DPPE-based lipid systems for nebulized antibiotics and inhaled biologics.
6. Gene and Cell Therapy Support:
Non-viral synthetic lipid systems enable transient, low-immunogenic gene delivery in ex vivo and in vivo settings. Examples include ionizable lipids used in CAR-T cell manufacturing and early-stage CRISPR LNP platforms.
Representative Clinical Findings
| Application | Delivery Route & Payload | Key Outcomes & Clinical Status |
| COVID-19 mRNA Vaccines | Intramuscular; mRNA encoding SARS-CoV-2 spike protein | >94% efficacy with strong immune response in Phase 3; marketed globally (Pfizer-BioNTech & Moderna)[1] |
| Onpattro (Patisiran) | Intravenous; siRNA targeting transthyretin (ATTR amyloidosis) | >94% efficacy with strong immune response in Phase 3; marketed globally (Pfizer-BioNTech & Moderna)[2] |
| mRNA-1944 & NTLA-2001 | Intravenous; mRNA therapeutics (antibody expression / gene editing) | Detectable RNA for >28 days with extended circulation; Phase 1/2 clinical evaluation[3] |
| Translate Bio MRT5005 | Inhalation; mRNA encoding CFTR | Generally safe and well tolerated; no significant FEV1 improvement; Phase 1/2 clinical[4] |
| Pulmonary mRNA LNP Dry Powder | Pulmonary; luciferase mRNA | Functional protein expression in lung and trachea; successful dry powder delivery in mice; preclinical[5] |
| Ocular mRNA LNP Delivery | Ocular; mRNA encoding lanosterol synthase | >7-fold higher delivery potency and reduced cataract severity in rats; preclinical[6] |
| Pulmonary Nebulized LNPs | Inhalation; CFTR mRNA | Sustained lung expression post-nebulization with no pulmonary or systemic toxicity; preclinical[7] |
Examples of Marketed Finished Products
| Product & Indication | Drug & Delivery System | Key Lipids | Photo |
| Spikevax (COVID-19) | mRNA · LNP | SM-102, DSPC, cholesterol, PEG2000-DMG | |
| Onpattro (hATTR amyloidosis) | siRNA · LNP | DLin-MC3-DMA, DSPC, cholesterol, PEG2000-DMG | |
| Vyxeos (AML) | Cytarabine + daunorubicin · Liposome | DSPC, DSPE, cholesterol | |
| Doxil / Caelyx (Breast & ovarian cancer) | Doxorubicin · PEGylated liposome | HSPC, cholesterol, PEG2000-DSPE | |
| AmBisome (Fungal infections) | Amphotericin B · Liposome | HSPC, DSPG, cholesterol | |
| Myocet (Breast cancer) | Doxorubicin · Non-PEG liposome | EPC, cholesterol | |
| DepoCyte (Lymphomatous meningitis) | Cytarabine · DepoFoam liposome | Phospholipids, cholesterol | |
| Onivyde (Pancreatic cancer) | Irinotecan · Liposome | DSPC, DSPE-PEG, cholesterol | |
| Visudyne (AMD) | Verteporfin · Liposome | Egg phosphatidylglycerol, DMPC | |
| Lipusu (NSCLC) | Paclitaxel · Liposome | Lecithin, cholesterol | |
| Exparel (Post-surgical analgesia) | Ropivacaine · DepoFoam liposome | Phospholipids, cholesterol | |
| Arikayce (NTM lung disease) | Amikacin · Inhaled liposome | DPPC, cholesterol |
Representative Customers using VAV’s Synthetic Lipids
| Market/Region | Customer Description | Focus Area |
| MEA | Middle Eastern Pharma Multinational | NDDS/Injectable |
| CIS | Regionally recognized biotech leader | Biologics & vaccines/Lipids nanoparticle |
| South Asia | One of the leading biotechnological company | Biologics & vaccines/Lipids nanoparticle |
| South America | Pioneer in scalable, sustainable bioscience | Vaccine adjuvants |
| LATAM America | Leading ophthalmic innovator | Ophthalmic |
| MEA | Innovator in medical devices | Medical Device |
| South Asia | Premier ocular healthcare producer | Ocular Disease/Injectable |
| South Asia | Pulmonary drug delivery innovator | Liposomal pulmonary delivery |
| South Asia | Pioneering research focused biotech | NDDS/Injectable |
| South Asia | Multinational biotechnology company | Lipid based nanoparticle |
| APAC | Prominent developer in ophthalmic therapeutics | Ophthalmic delivery |
| CIS | Top-tier Russian lifescience company | Diagnostic products |
| APAC | Globally operating pharmaceutical CDMO | Liposomal drug delivery/Injectable |
| APAC | Specialty life sciences company | NDDS/Injectable |
Conclusion
Synthetic lipids have established themselves as a core enabling technology for modern drug and biologics delivery, supporting advances across mRNA therapeutics, gene editing, RNA interference, and targeted oncology. Their molecular tunability, biocompatibility, and formulation flexibility have expanded what is achievable with complex and sensitive therapeutic payloads.
Clinical and commercial successes across vaccines, rare diseases, oncology, and localized delivery routes have validated synthetic lipid platforms as reliable, scalable, and adaptable solutions. As therapeutic modalities continue to evolve, synthetic lipids will play an increasingly central role in bridging innovative science with real-world patient impact.
Sustained progress in this field will be driven by access to high-quality, customizable lipid technologies and partners capable of supporting development from early research through commercialization. VAV Lipids brings together GMP-grade synthetic lipids, deep technical expertise, and a collaborative approach to help translate next-generation therapies into clinical and commercial reality.
References
- NCT04470427. Efficacy, safety, and immunogenicity of mRNA-1273 vaccine in adults ≥18 years for COVID-19 prevention.
- Urits I, et al. Patisiran (ONPATTRO) for hereditary transthyretin amyloidosis polyneuropathy. Neurol Ther. 2020;9:301–315.
- Suzuki Y, et al. Intravenous lipid nanoparticles in clinical trials: links to extracellular vesicles. Mol Pharm. 2023;20(10):4883–4892.
- NCT03375047. Safety and tolerability of nebulized MRT5005 in adults with cystic fibrosis (RESTORE-CF).
- Sarode A, et al. Inhalable dry-powder mRNA lipid nanoparticles for pulmonary delivery. Drug Deliv Transl Res. 2024;14(2):360–372.
- Song R, et al. Ocular delivery of LNP-mRNA encoding lanosterol synthase for cataract treatment in rats. Nat Commun. 2025;16:8522.
- Kim J, et al. Lipid nanoparticle interactions and intracellular delivery mechanisms. ACS Nano. 2022;16(9):14792–14806.
