Executive Summary
The skin’s complex barrier often limits the effectiveness of topical treatments. Liposomes, nanoscale phospholipid vesicles, offer a breakthrough by enabling deeper, targeted, and controlled delivery of cosmetic actives. They serve dual roles: restoring the skin barrier and acting as carriers that encapsulate both hydrophilic and lipophilic compounds.
This whitepaper highlights their advantages—enhanced penetration, improved stability, sustained release, and reduced irritation—backed by case studies on actives like caffeine, salicylic acid, and bakuchiol. Results show superior hydration, oil control, pore refinement, and wrinkle reduction compared to conventional formulations.
By combining biocompatibility with precision delivery, liposomes elevate skincare efficacy and safety. They represent the future of cosmetic innovation, offering consumers longer-lasting, more effective, and science-driven solutions.
Introduction to Skin
The skin, the body’s largest organ, covers about 2 m² and makes up 12–15% of body weight. It protects against injury, pathogens, UV radiation, and chemicals while regulating hydration and temperature. Structurally, it has three layers: the epidermis, providing barrier protection and vitamin D synthesis; the dermis, rich in collagen, vessels, and nerves for strength, elasticity, and sensation; and the hypodermis, a fatty layer offering cushioning, insulation, and energy storage.
This multilayered architecture makes topical and transdermal delivery difficult. Liposomes, with their biocompatibility and ability to carry both water- and fat-soluble actives, overcome these barriers through improved penetration, stability, and controlled release.
Introduction to Liposomes
Liposomes are nanoscale vesicles composed of phospholipids that closely resemble natural cell membranes. Their unique bilayer structure enables the encapsulation of both water- and fat-soluble actives, shielding them from degradation and enhancing stability. This structural versatility makes liposomes highly effective carriers, improving bioavailability and enabling targeted delivery into deeper layers of the skin.
Their properties can be tailored for specific applications: smaller liposomes penetrate more effectively, while positively charged ones interact strongly with negatively charged skin cells. By fusing with the skin’s lipid layers, liposomes enhance penetration, retention, and sustained release, while minimizing irritation and systemic absorption.
Key advantages include: biocompatibility, protection of sensitive ingredients, versatile encapsulation, controlled delivery, and improved therapeutic performance- making liposomes a transformative technology in advanced skincare formulations.
Liposomes in Skincare: As Active Agents for Skin Barrier Repair
Beyond serving as carriers, empty liposomes exhibit therapeutic properties of their own. In compromised skin conditions such as eczema or dehydration, they interact directly with skin lipids, proteins, and carbohydrates. This interaction aids in the reconstruction of the skin barrier, helping the stratum corneum restore its integrity and regain its protective function.
Liposomes in Skincare: As Advanced Delivery Vehicles
As delivery systems, liposomes provide multiple benefits that elevate the performance of active ingredients:
- Penetration: Improve absorption of active compounds into deeper skin layers.
- Solubility: Facilitate delivery of water- or oil-insoluble ingredients.
- Stability: Protect sensitive ingredients from degradation due to light, air, or pH.
- Controlled Release: Prolong the effects of active compounds over time.
- Pharmacokinetics & Pharmacodynamics: Enable site-specific action for more efficient treatment.
- Reduced Toxicity: Minimize side effects by limiting systemic exposure.
1. Penetration
The skin, the body’s largest organ, acts as a protective barrier with three layers: epidermis, dermis, and hypodermis. The stratum corneum, the outer epidermis, is the main barrier due to its lipophilic nature. Few substances penetrate it effectively, limiting active ingredient delivery. Liposomes, mimicking skin lipids and being nanosized, enhance stratum corneum penetration better than conventional formulations.
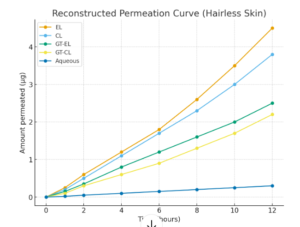
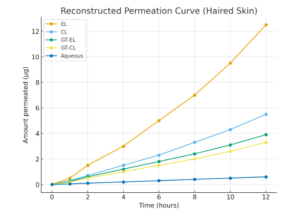
Case Study: Elastic liposomes (EL) were compared with conventional liposomes (CL) for delivering GT across haired and hairless skin using in-vitro Franz diffusion cells.
Key Findings
- Higher Drug Loading: ELs encapsulated more GT due to sodium deoxycholate, which improves solubility and membrane flexibility.
- Greater Flexibility: ELs were 3.5× more deformable, aiding passage through skin layers.
- Improved Penetration & Accumulation: ELs enhanced GT permeation and deposition in haired skin, likely via follicular pathways.
- Role of Hair Follicles: Benefits were reduced in hairless skin, showing follicles are key for EL-mediated delivery.
Clinical Conclusion: Elastic liposomes enhance GT transdermal delivery, especially via hair follicles, improving drug loading, penetration, and therapeutic efficacy. In haired skin, EL enhances GT permeation and deposition over CL, whereas in hairless skin, both perform similarly.
2. Solubility
Liposomes’ biphasic structure allows encapsulation of hydrophilic, amphiphilic, and lipophilic molecules—hydrophilic agents in the aqueous core and lipophilic/amphiphilic compounds in the lipid bilayer—reducing material loss and enabling delivery of hydrophobic substances in aqueous systems.
Case Study: Caffeine delivery via liposomes was influenced by vesicle properties modified with edge activators or penetration enhancers.
Key Findings
- Smaller Size & Flexibility: Propylene glycol increased
- bilayer fluidity, producing smaller, more deformable vesicles for better skin penetration.
- Enhanced Transdermal Delivery: Optimized liposomes improved caffeine absorption through flexibility and skin interaction.
- Mechanisms: Liposome fusion with stratum corneum, lipid matrix perturbation, and direct solute transfer.
- Safety: HaCaT keratinocyte studies showed no cytotoxicity below 3.3% v/v.
Clinical Conclusion: Propylene glycol-modified liposomes boost hydrophilic drug delivery safely, enhancing bioavailability in topical formulations.
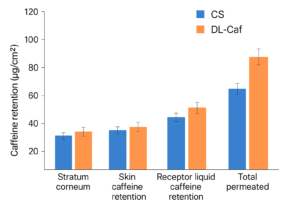
Comparison of Permeation Parameters: Control Solution vs. Deformable Liposomal Caffeine formulation
3. Stability
Many active substances are vulnerable to oxidation, degradation, or reduced efficacy due to environmental factors. Liposomes can protect these ingredients by encapsulating them. While the skin has its own antioxidant defense system, UV exposure can trigger oxidative stress and photoaging. To combat this, topical antioxidant supplementation is widely used, though many antioxidants are unstable. Encapsulating them in liposomes helps preserve their activity and enhance skin delivery.
4. Controlled Release
Current cosmetic research focuses on nanoparticulate systems for controlled skin delivery, with liposomes being widely recognized due to their similarity to the stratum corneum. Topical liposomes deposit lipids into the skin, enabling gradual release and prolonged action, mostly in the upper stratum corneum.
Case Study: Salicylic acid was encapsulated in liposomes to achieve sustained release, reduced irritation, and enhanced skin retention for dry skin and acne.
Key Findings
- Controlled Release: Optimized liposomes (0.35 µm) released 85.7% of salicylic acid in 6 hours, slower than other batches (>90%).
- Improved Retention: Liposomes showed 65.96% skin retention vs. 6.15% for plain dispersion (p<0.05).
- Formulation Benefits: Lecithin and propylene glycol improved moisturization and entrapment efficiency (42.6%).
Clinical Conclusion: Liposomal encapsulation enhances sustained release, skin retention, and bioavailability of salicylic acid while reducing irritation, offering promise for cosmeceutical applications.
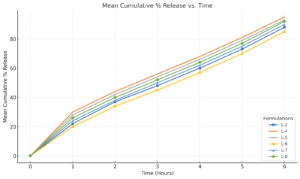 Release profiles of salicylic Release profiles of salicylic acid from liposomes
Release profiles of salicylic Release profiles of salicylic acid from liposomes
5. Pharmacokinetics & Pharmacodynamics
Due to the skin’s low permeability, many substances require frequent application without a delivery system. Liposomes enhance the pharmacokinetics of cosmetic products by prolonging therapeutic effects, improving targeting specificity, and reducing toxicity. They can also be formulated for extended circulation and sustained ingredient release.
6. Reduced Toxicity
Liposomes act as protective carriers, isolating active ingredients to prevent unwanted interactions and formation of toxic compounds. They enable targeted delivery, controlled release, and lower effective doses, reducing the risk of toxicity.
Case Study: Liposome-encapsulated 0.5% bakuchiol cream was compared with conventional 0.5% bakuchiol oil.
Key Findings
- Hydration: Encapsulated cream increased moisture by 11% vs. 4% for oil.
- Pore Reduction: 33% vs. 10% (p=0.005).
- Sebum Control: 41.5% vs. 33% (p=0.003).
- Wrinkle Reduction: 76% vs. 54% (p=0.001).
Mechanism & Stability: Gradual release, deeper penetration, and protection from degradation enhanced efficacy. Encapsulated cream showed superior emulsion stability, pH, and spreadability.
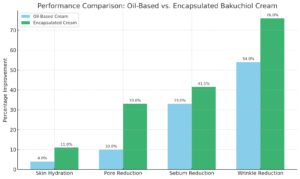
Performance comparison: Oil based vs. Encapsulated Bakuchiol cream
Clinical Conclusion: Liposomal 0.5% bakuchiol cream outperformed oil-based bakuchiol in hydration, pore size, sebum control, and wrinkle reduction. Liposomal delivery enhances topical efficacy, skin tolerance, and stability, making it ideal for long-term and sensitive-skin use.
Characterization of Liposomes
Particle size:
- Smaller the particle size, better absorption.
- Ideal size of liposomes – 200 – 500 nm.
- Ensures better absorption
Polydispersity Index (PDI):
- Measure of width of unimodal size distribution
- Acceptable PDI should have the value < 0.7
- The PDI of the liposomes indicate dispersion homogeneity
Electron Microscopy
Examples of Marketed Liposomal Products for Skin Delivery
| Customer Description | Liposomes & Key Ingredients | Region,
Market |
| High-end skincare, perfumes producer | Liposomes in Gel | Western Europe |
| Global leader in beauty & personal care | Tanning agents in Liposomes | Western Europe |
| Global American company in innovative skincare | TMF, vitamins E, A palmitate, cerebrosideceramide | Northeastern United States |
| Leading Nanotechnology based skincare manufacturer | Antipigmentation agents in liposomal serums | Southwestern Europe |
| High performance skincare formulator | Antiwrinkle agents in liposomal cream | Northwestern India |
Conclusion
Liposomal active ingredients represent a promising advancement in enhancing the delivery and effectiveness of skincare formulations. Owing to their unique structure and functional properties, liposomes can encapsulate and protect active compounds, facilitate deeper penetration into the skin, and enable targeted delivery to specific skin layers or cells.
As scientific research continues to expand in this area, we can anticipate the emergence of increasingly innovative and high-performance cosmetic products leveraging this advanced delivery system. The future of skincare is poised for significant progress with the continued evolution of liposomal technology.
References
- Nastruzzi, C., Esposito, E., Menegatti, E., & Walde, P. (1993). Use and stability of liposomes in dermatological preparations. J Appl Cosmetol, 11, 77–91.
- Kyoung Hoon Kang a., Myung Joo Kang a., & Jaehwi Lee a. (2010). Influence of Liposome Type and Skin Model on Skin Permeation and Accumulation Properties of Genistein. J. Dispersion Sci. Technol, 31:1061–1066.
- Li, S., & Young Wook Choi (2011). Enhanced transdermal delivery of 18β-glycyrrhetic acid via elastic vesicles: in vitro and in vivo evaluation. Drug Dev Ind Pharm, 37(9), 1035–1044.
- Abd E, Gomes J, Sales CC, Yousef S, Forouz F, Telaprolu KC, et al. (2020). Deformable liposomes as enhancer of caffeine penetration through human skin in a Franz diffusion cell test. Int J Cosmet Sci, 43(1), 1–10.
- Bhalerao, S. S., & Harshal, A. R. (2003). Preparation, optimization, characterization, and stability studies of salicylic acid liposomes. Drug Dev Ind Pharm, 29(4), 451–467.
- Chatterjee, K., Kumar, A., Mitra, A., & Mitra, S. (2022). Comparative efficacy of bakuchiol oil and encapsulated bakuchiol cream on facial skin quality: A 28-day pilot study. J Cosmet Dermatol, 21(3), 1062–1070