Executive Summary
Egg phospholipids (lecithin) are well established pharmaceutical excipients with more than five decades of safe clinical use in injectable emulsions and parenteral formulations. Their amphiphilic structure enables effective stabilization of oil-in-water emulsions, solubilization of poorly water soluble drugs, and improved bioavailability, while maintaining excellent biocompatibility & broad global regulatory acceptance.
They address key challenges in injectable drug delivery, including formulation instability, toxicity risks, and patient safety by enabling fine emulsions with controlled droplet size & reproducible performance. Their clinical utility is demonstrated in propofol anesthetic emulsions, lipid-based amphotericin B formulations with reduced nephrotoxicity, and total parenteral nutrition emulsions.
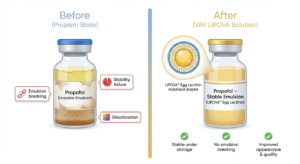
cGMP manufactured egg phospholipids from VAV provide consistent quality, batch reproducibility, and strong regulatory support, reinforcing their continued importance in modern injectable drug delivery systems.
Introduction
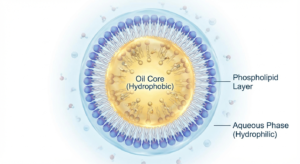
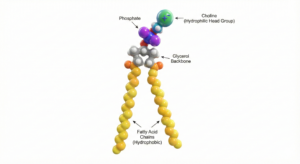
Phospholipids are amphiphilic molecules consisting of a hydrophilic phosphate head group and hydrophobic fatty acid tails. This dual nature enables their unique ability to stabilize oil-in-water emulsions by orienting themselves at the interface between aqueous and lipid phases.
The pharmaceutical industry has relied on egg phospholipids for over five decades as a primary excipient for parenteral formulations. Their acceptance by regulatory authorities worldwide including the U.S. Food and Drug Administration (USFDA), European Medicines Agency (EMA), and Japanese Pharmaceuticals and Medical Devices Agency reflect their established safety profile and efficacy.
Challenges in Lipophilic Drug Delivery
Lipophilic drugs pose major challenges in parenteral formulation. Poor aqueous solubility (often LogP >3) leads to low and variable bioavailability. Without appropriate formulation, these drugs are prone to aggregation or precipitation, causing instability. Improper formulation may also increase toxicity, particularly hepatic and renal effects. Ensuring emulsion stability during storage and dilution is critical to patient safety.
Egg Phospholipids: The Solution
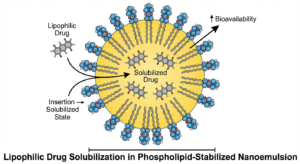
Egg phospholipids overcome these challenges through surface-active emulsification, forming stable oil-in-water droplets (typically 100–500 nm). They enhance solubility by incorporating lipophilic drugs into lipid phases, enable controlled release through phospholipid composition, and offer excellent biocompatibility by mimicking biological membranes. Decades of parenteral use support their strong regulatory acceptance.
Chemistry and Composition
Mechanisms of Action
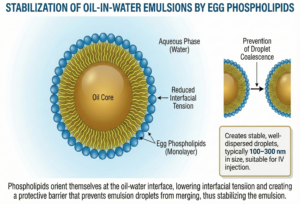
Emulsification and Interfacial Stabilization
Egg phospholipids function as emulsifiers by accumulating at the oil-water interface, reducing interfacial tension and preventing coalescence of emulsion droplets.
The phosphate head group orients toward the aqueous phase (hydrophilic), while the fatty acid tails orient toward the oil core (hydrophobic). This structure creates a stable barrier that prevents droplet fusion
Particle Size Distribution and Stability
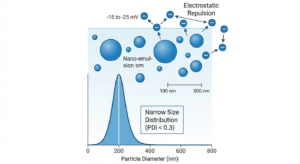
The particle size of emulsion droplets is critical to formulation stability and efficacy. Egg phospholipids enable the formation of fine emulsions with:
- Mean droplet diameter: 100 to 500 nm (depending on formulation and processing)
- Polydispersity Index (PDI): <0.3 (indicating narrow, uniform distribution)
- Zeta potential: -15 to -25 mV (conferring electrostatic stability).
The negative zeta potential arises from ionizable phosphate groups on the phospholipid surface, creating repulsive forces between droplets and preventing aggregation.
Solubilization of Lipophilic Drugs
Egg phospholipids improve the solubility of lipophilic drugs by enabling their incorporation into the lipid core of stabilized oil-in-water emulsion droplets. This maintains drugs in a solubilized state, prevents precipitation, and enhances aqueous dispersion, resulting in improved bioavailability.
Key Applications
1. Propofol Injection
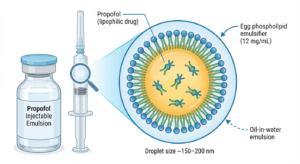
Clinical Significance: Propofol is one of the most widely used intravenous anesthetics worldwide, with annual consumption exceeding 500 metric tons.
Formulation Challenge: Propofol is highly lipophilic (LogP≈4.0) and practically insoluble in water, preventing conventional aqueous injectable formulations. mg/mL, forming stable oil-in-water emulsions with mean droplet sizes of 150-200 nm.
Clinical Benefits: This formulation enables smooth IV administration, consistent pharmacokinetics, broad compatibility with IV fluids, and excellent physical stability.
Stability Profile: Propofol emulsions remain stable for 24-36 months unopened, several hours after opening, and up to several days after dilution, provided pH is maintained within the optimal range of 6.0-8.5.
2. Amphotericin B Injectable Emulsion
Clinical Significance: Amphotericin B is a cornerstone therapy for invasive fungal infections.
Formulation Challenge: Extremely poor aqueous solubility and high nephrotoxicity of conventional deoxycholate formulations necessitate lipid-based delivery.
Egg Phospholipid Solution: Egg phospholipids serve as primary emulsifiers in amphotericin B emulsions, enabling safe parenteral delivery.
Clinical Benefits: Phospholipid-based emulsions reduce nephrotoxicity and infusion reactions, improve tolerability, and maintain antifungal efficacy.
3. Total Parenteral Nutrition (TPN)
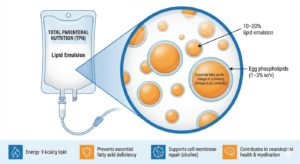
Clinical Significance: Total parenteral nutrition is essential for patients with non functional gastrointestinal tracts, providing complete nutritional support.
Role of Egg Phospholipids: Egg phospholipids act as emulsifiers in 10-20% lipid emulsions, typically at 1-3% (w/v), enabling stable delivery of essential fatty acids, including linoleic and α-linolenic acids.
Nutritional Contribution: Lipid emulsions provide a dense energy source and prevent essential fatty acid deficiency in long-term TPN. Phospholipids also supply choline, supporting cell membrane integrity and neurological function.
Examples of Marketed Finished Products (Formulations) with Egg Phospholipids
| Brand | Region | Active & Therapeutic Category | Photo |
| Diprivan | Worldwide | Propofol 1%, Anesthetic | |
| Amphomul | Asia | Amphotericin B 0.5%, Anti Fungal | |
| Cinvanti | USA, Europe | Aprepitant, Antiemetic | |
| Diazemuls | Europe, CA, AU | Diazepam, Anxiolytic Sedative/Anticonvulsant | |
| Cleviprex | USA | Clevidipine Butyrate, Antihypertensive | |
| Intralipid | Worldwide | Emulsion, Parenteral
Nutrition |
|
| Lipofundin
MCT/LCT |
Europe, Asia | Emulsion, Parenteral
Nutrition |
Representative Customers using VAV’s Egg Phospholipids (Lecithin)
| Company Profile | Market / Region | Application |
| Leading Anesthetic Manufacturer | Latin America | Propofol injectable emulsion |
| Major Parenteral Nutrition Producer | Balkan | TPN lipid emulsion |
| Regional Anesthetics Manufacturer | CIS | Propofol injectable emulsions |
| Veterinary Pharmaceutical Company | CIS | Compounded veterinary sedative injections |
| Top Generic Injectables Manufacturer | Asia | Propofol injectable emulsions |
| Leading Injectables CDMO | Asia | TPN lipid emulsion |
Conclusion
Egg phospholipids represent one of the most successful, versatile, and clinically validated pharmaceutical excipients, with a history spanning multiple decades of safe use in parenteral formulations. Their unique amphiphilic properties enable the solubilization and stabilization of lipophilic drugs, creation of injectable emulsion delivery systems, and support of critical parenteral nutrition.
The science underlying phospholipid-based formulations continues to advance, with new applications emerging in oncology, gene therapy, and vaccine development. The proven safety profile, regulatory acceptance, and consistent supply of pharmaceutical-grade egg phospholipids make them an indispensable component of modern drug delivery innovation.
References
- Calder PC, Jensen GL, Koletzko BV, Singer P, Wanten GJ. (2010). “Lipid emulsions in parenteral nutrition of intensive care patients: Current thinking and future directions.” Intensive Care Medicine, 36(5), 735-749.
- Husna M, Noviyanto A, Permatasari DA, Insani NN. (2021). “Systematic review of the stability and compatibility of propofol emulsion in various storage conditions and containers.” Journal of Pharmaceutical and Biomedical Analysis, 197, 113912.
- Brüggemann RJ, Alffenaar JW, Blijlevens NM, et al. (2022). “Liposomal amphotericin B- the past, present, and future.” Journal of Antimicrobial Chemotherapy, 77(Supplement_2), ii3-ii13.
- Seifert SA. (2016). “Nephrotoxicity of amphotericin B formulations: a comparative review.” Current Fungal Infection Reports, 10(3), 95-102.
- Collins-Gold L, Szoka FC Jr. (1990). “Parenteral emulsions for drug delivery.” Advanced Drug Delivery Reviews, 5(3), 189-208.
- Fruhwirth GO, Hermetter A. (2007). “Impacts of phospholipids on nutrition and health.” Molecular Nutrition & Food Research, 51(11), 1385-1394.
- Phospholipid Research Center. (2020). “Current Research in Phospholipids and Their Use in Drug Delivery.” Lipids in Health and Disease, 19, 123.
